Radiochemistry Discovery and Multifunctional Nanomaterials
Radiochemistry Discovery
The overall research goal of the Radiochemistry Program is to synthesize novel positron emission tomography (PET) labeled compounds and radiopharmaceuticals for preclinical and clinical evaluations.
Our team has expanded to include faculty members, visiting scientists, research associates, postdoctoral fellows, undergraduate and high school students. We also have an active clinical production team that translates new PET radiopharmaceuticals for clinical studies.
List of Available Agents for Human and Pre-Clinical Use at MGH PET Core
List of Available Agents for Human and Pre-Clinical Use at MGH PET Core
Available for Clinical Use at MGH PET Core
F-18
- FDG
- FES (estrogen receptor)
- T807 (tau protein)
- FPEB (mGlur5)
- BFPET (myocardial perfusion)
- GE180 (TSPO/PBR)
- FLT (cellular proliferation)
C-11
- PiB (amyloid)
- Altropane (Dopamine transport)
- Raclopride (D2/D3 receptor imaging)
- Methionine (amino acid update)
- Acetate (fatty acid synthase)
- CO (gas)
- Rolipram (PDE-4 receptor imaging)
- P943 (5-HT1B)
N-13
- Ammonia
- N2 in Saline (lung perfusion imaging)
O-15
- C15O2 (blood flow imaging)
Available for Pre-Clinical Use at MGH PET Core
F-18
- THK 5351/GE-216 (tau protein)
- FMISO
- MK-6240 (tau protein)
- GE179 (NMDA)
- Fallypride
- FHBG
- FACBC
- Fluorocholine
- Fluoroacetate
- Fluoroethyl tyrosine (FET)
- NaF
- F-18 target material
C-11
- PHNO
- PBR-28 (TSPO/PBR)
- (S,S)-[11C]Methylreboxetine (MeNR/MRB) – targets NET
Multifunctional Nanomaterials in Biology (MNB)
This research area was led by Dr. Lee Josephson who has over 100 peer-reviewed publications and 30 issued US patents. He is a co-founder of AMAG Pharmaceuticals and T2 Biosystems, a company developing MR-based assays and sensors.
The main research topics are (i) magnetic nanoparticles as MR contrast agents and for MR-based bioassays, (ii) multifunctional nanoprobe chemistries, (iii) imaging probes of cell stress and cell death induced by ischemia and cancer chemotherapy.
(i) Magnetic Nanoparticles: We have attached DNA binding fluorochromes to nanoparticle (NP) surfaces, to obtain DNA binding NP’s that can detect PCR generated DNA or be used for imaging the DNA in biological systems. In collaboration with Dr. David Sosnovik, we have attached annexin V to NP’s and demonstrated that annexin-NP’s use can be used to image ischemia induced apoptosis or cell death. Annexin-NP’s exhibit a strong protective effect against plasma cell rupture and can decrease infarct size in animal models. With their diagnostic and therapeutic applications, Annexin-NP’s provide a broad, clinically translatable approach to theranostic NP’s.
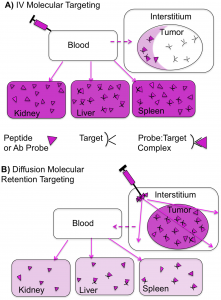
(ii) Multifunctional PEG-like Nanoprobes: We react PEG polymers with short peptides, to yield protein-sized, molecularly targeted, or passively targeted fluorescent nanoprobes featuring a radiolabeling option. These PEG-like Nanoprobes (PN’s) can be used for a highly efficient, molecular targeting termed Diffusion Molecular Retention (DMR) tumor targeting. DMR bypasses the numerous barriers between the vascular compartment and tumor interstitium encountered with IV injection, by injecting PN’s interstitially and allowing PN diffusion through the normal and tumor interstitium to the tumor, with extremely low levels of liver and kidney uptake. PN’s can also be used as a broad platform for optimizing, by PEG variation, the pharmacokinetics of passively targeted diagnostic and therapeutic agents.
(iii) Imaging probes for cell death: We have employed a variety of annexin V-based probes (bind apoptotic and necrotic cells), cathepsin B probes (bind autophagic cells), and DNA binding fluorochromes (bind necrotic cells). Our long-term goal is the development of panel of probes that can be used to image the various phases of cell death (apoptosis, necrosis, autophagy) in fluorescent, cell-based research or SPECT or PET imaging.
Magnetic Nanoparticles
From a recent ACS Nano paper entitled Fluorochrome-Functionalized Nanoparticles for Imaging DNA in Biological Systems:
Attaching DNA binding fluorochromes to nanoparticles (NPs) provides a way of obtaining NPs that bind to DNA through fluorochrome mediated interactions. To obtain a nanoparticle (NP) that bound to the DNA in biological systems, we attached the DNA binding fluorochrome, TO-PRO 1 (TO), to the surface of the Feraheme (FH) NP, to obtain a fluorochrome-functionalized NP denoted TO-FH. When reacted with DNA in vitro, TO-FH formed microaggregates that were characterized by fluorescence, light scattering, and T2 changes.
Reprinted with permission from Fluorochrome-Functionalized Nanoparticles for Imaging DNA in Biological Systems, Hoonsung Cho, David Alcantara, Hushan Yuan, Rahul A. Sheth, Howard H. Chen, Peng Huang, Sean B. Andersson, David E. Sosnovik, Umar Mahmood, and Lee Josephson, ACS Nano 2013 7 (3), 2032-2041, Copyright 2013, American Chemical Society.
High Efficiency Diffusion Molecular Retention Tumor Targeting
From a recent PLOS ONE paper:
Diffusion molecular retention (DMR) tumor targeting is a technique that employs PEG-fluorochrome shielded probes that, after a peritumoral (PT) injection, undergo slow vascular uptake and extensive interstitial diffusion, with tumor retention only through integrin molecular recognition.

Yanyan Guo, Hushan Yuan, Hoonsung Cho, Darshini Kuruppu, Kimmo Jokivarsi, Aayush Agarwal, Khalid Shah, Lee Josephson, “High Efficiency Diffusion Molecular Retention Tumor Targeting”, 2013, PLoS ONE 8(3): e58290. doi: 10.1371/journal.pone.0058290 [Link]